Apr
2022
Thermal Safety Design of New Energy Battery Systems
13 Apr 2022
This article aims to analyze the importance of thermal safety design, market thermal safety issues, solutions for battery system thermal safety design, and thermal runaway warning strategies. It is intended to serve as a reference for implementing corresponding protective measures in the thermal safety design of power batteries.
Importance of Thermal Safety Design
With the increasing number of electric vehicles in circulation, safety incidents involving electric vehicles have significantly risen and become a focal point of concern.
The operation of power batteries involves a complex electrochemical reaction process. Under conditions of high temperature, high pressure, high current, and large currents, incomplete electrochemical reactions may occur, leading to localized overheating and triggering thermal runaway.
If effective control measures are not promptly implemented after a battery experiences thermal runaway, the substances generated from internal reactions will further propagate, ultimately resulting in explosions and causing casualties.
Therefore, studying the heat generation mechanisms and control strategies during the thermal runaway process of batteries is of vital significance in preventing and reducing electric vehicle fire accidents.
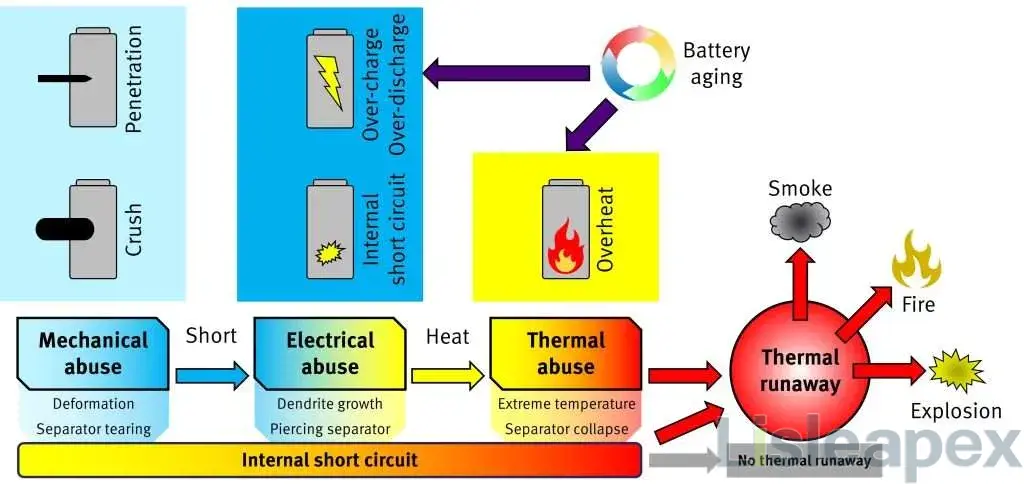
Principles of Thermal Runaway and Propagation
Thermal runaway in power batteries can be caused by internal and external factors.
Internal factors generally include overcharging, low-temperature charging, negative electrode defects leading to compound penetration through the separator and causing short circuits, or impurities piercing the separator within the battery.
External factors encompass short circuits between positive and negative electrodes, high current discharges, high temperatures, compression, and punctures.
As a result of internal or external factors occurring, the temperature of battery cells continues to rise.
Taking lithium-ion batteries as an example, when the cell temperature exceeds 60°C, the solid electrolyte interface (SEI) film begins to decompose, exposing the surface of the negative electrode.
With further temperature increase, the battery separator contracts due to high temperatures, resulting in contact between the positive and negative electrode active materials, leading to a short circuit and instantaneous release of a large amount of heat.
The high-temperature short circuit point further causes the decomposition of the positive electrode oxide, releasing free oxygen that reacts with the organic electrolyte, thereby releasing more heat. This eventually leads to the ignition and explosion of the battery.
When an internal short circuit occurs in a lithium-ion battery, the immense Joule heat causes the less stable solid electrolyte interface (SEI) film to decompose.
The initial temperature ranges from 80 to 120°C, and the reaction equation is as follows:
(CH2OCO2Li)2 → Li2CO3 + C2H4 + CO2 + 0.5O2
2Li + (CH2OCO2Li)2 → 2Li2CO3 + C2H4
After the decomposition of the SEI film, the protective properties of the negative electrode active material are lost.
The embedded lithium within it comes into contact with the electrolyte, resulting in an exothermic reaction.
At the same time, a relatively sparse SEI film is formed again, but it has no impact on the overall reaction.
The reaction temperature at this stage ranges from 120 to 250°C, and the reaction equations are as follows:
2Li2 + C3H4O3 → Li2CO3 + C2H4
2Li + C5H10O3 → Li2CO3 + C2H4 + C2H6
2Li + C3H16O3 → Li2CO3 + C2H6
2Li + C4H6O3 → Li2CO3 + C3H6
Under the continuous exothermic reaction between the negative electrode and the electrolyte, the separator in the battery shrinks and breaks, allowing further contact between the positive and negative electrode active materials, resulting in more heat generation and further collapse of the separator.
Subsequently, the positive electrode material starts to decompose, and the generated oxygen reacts violently with the electrolyte.
Along with reactions involving the electrolyte, binders, and outer packaging, the temperature rise rate increases sharply, entering a state of thermal runaway.
In summary, when a lithium-ion battery is subjected to a needle puncture, an intense internal short circuit occurs within a short period.
When the battery casing cannot withstand the pressure caused by the gas and heat, the internal chemical substances will be ejected instantly, leading to smoke, fire, and even explosions.
Thermal Safety Design of Battery Systems
The development of thermal safety design primarily involves five dimensions: cell selection, module design, battery system thermal safety design, thermal runaway warning, and active cooling.
Cell Design and Selection
Cell Selection: High thermal stability, improved shell strength, reduced vent valve pressure, enhanced insulation.
Cell design is the core of battery system design. Only with scientifically rigorous cell design can problems and failures be minimized during later stages of application. There are several key points in cell design, and achieving the desired results requires a balanced design from multiple perspectives.
Common cell manufacturing processes include stacking and winding. Stacking involves pressing layers together, while winding is the process of layering them externally.
Module Design
Module design mainly involves bridging the actual conditions of individual cells with the requirements of the pack.
It includes structural design requirements, thermal design requirements, electrical design requirements, and safety design.
Structural design requirements: Reliability (vibration resistance, fatigue resistance), controllable manufacturing processes (no excessive or virtual welding), ensuring 100% cell integrity.
Thermal design: The physical structure of pouch cells makes them less prone to explosion.
Generally, an explosion is only possible if the shell can withstand sufficient pressure.
When the internal pressure of a pouch cell increases, it will release pressure and leak from the edges of the aluminum-plastic film.
Pouch cells also have the best heat dissipation among different cell structures.
Electrical design: Low-voltage design involves using signal acquisition harnesses to collect battery voltage and temperature information to the module control board or the so-called module controller mounted on the module.
High-voltage design focuses on the series-parallel connection between cells and the connection and conduction methods between modules. Typically, only series connections are considered between modules.
Safety design: Safety design can be divided into three backward requirements: ensuring a well-designed system to prevent accidents;
if an accident does occur, it is best to provide early warning to allow for response time; if a failure has already occurred, the design objective shifts to preventing the rapid propagation of accidents.
Thermal Runaway Warning Strategy
We divide the warning system into two parts: vehicle-side and cloud-side. On the vehicle-side, we integrate the temperature, pressure, and smoke signals within the battery pack and combine them with voltage, temperature, and other factors that can lead to thermal runaway.

These signals are then transmitted to the Vehicle Control Unit (VCU) or the Thermal Management System (TMS).
The overall vehicle thermal management system will respond to these signals and initiate a rapid cooling process.
It is crucial to consider the coolant volume in the new energy vehicle and the timing of cooling activation.
Conclusion
Significant progress has been made in the study of individual cell thermal runaway, but safety incidents still occur worldwide. Further research and practical verification are required for the grouped technology of battery systems.
In the product development process, it is essential to validate the safety performance through thermal runaway safety tests and confirm the effectiveness of the thermal runaway safety design.
Recommended Industry
-
Utilizing High Ripple Current for Smarter Electric Vehicle Designs
According to the latest research by market analysis firm Markets and Markets, the electrification of the automotive industry is projected to grow at a compound annual growth rate of 11.9%, increasing from $73.7 billion to $129.6 billion by 2025. This rapid growth also signifies substantial investments in research and development as electric vehicles continue to gain widespread consumer acceptance.
-
Thermal Safety Design of New Energy Battery Systems
This article aims to analyze the importance of thermal safety design, market thermal safety issues, solutions for battery system thermal safety design, and thermal runaway warning strategies. It is intended to serve as a reference for implementing corresponding protective measures in the thermal safety design of power batteries.
-
Improving Visibility with DLP® Headlights
Automotive manufacturers are seeking methods to enhance nighttime driving visibility. DLP® automotive technology, applicable to headlights, can improve visibility and support other applications.
Stay updated with Lisleapex by signing up for the newsletter


 Congratulations On Your Successful Submission
Congratulations On Your Successful Submission
 Submission Failure
Submission Failure